Latest Update on NCQA Kidney Health Evaluation for Patients With Diabetes (KED) Measure Specification: Conformant Tests
June 4, 2024 · Eric Schneider, MD, M.Sc.
In August 2023 NCQA removed a LOINC code for semi-quantitative testing from the Kidney Health Evaluation for Patients With Diabetes (KED) HEDIS® measure specifications starting in Measure Year 2024.
We have received questions about removing the LOINC code. This page contains the latest information on appropriate urine albumin-creatinine ratio (uACR) testing for the KED HEDIS measure.
What is the purpose of the KED measure?
The KED measure looks at the percentage of members 18–85 years of age with diabetes (type 1 and type 2) who received a kidney health evaluation, defined by an estimated glomerular filtration rate (eGFR) and a urine albumin-creatinine ratio (uACR), during the measurement year. The measure’s intent is to monitor kidney health among adults with diabetes to help identify and prevent progression of chronic kidney disease. The measure is not intended to be a broad screening measure.
What type of testing is specified for this measure?
Guidelines indicate that quantitative blood and urine tests are to be used in combination to diagnose chronic kidney disease and to score a patient’s risk. Guidelines indicate that semi-quantitative urine tests performed at home or at the point of care can be useful for screening, but are expected to be confirmed by a quantitative urine test performed in an accredited laboratory. In line with this expectation that a quantitative test will be performed, the quality measure specification includes only quantitative tests. A semi-quantitative test performed alone (without a confirmatory quantitative test) is not considered adequate for evaluating and managing kidney disease.
What is the difference between quantitative and semi-quantitative tests, and why is only quantitative testing specified?
There are different types of uACR urine tests:
- Quantitative test results are reported as a numeric value. They are used in combination with a blood test to diagnose chronic kidney disease, and can be used to monitor the progression of disease.
- Semi-quantitative test results are reported in categories or ranges. They are less precise, but can be done rapidly at home or in an office. Patients can learn whether their results are in an abnormal range, but not exactly how far from normal they are. Like other semi-quantitative tests, they can be used to detect a potential problem that warrants follow-up. Because they are less precise, semi-quantitative tests typically require confirmation by a quantitative test processed by an accredited laboratory.
Clinical guidelines for chronic kidney disease include an algorithm that takes a quantitative value from a blood test and a quantitative value from a urine test and assigns the patient to one of six risk categories. A patient’s risk category helps determine the best treatment options and most effective care. Below is an example of a risk categorization chart commonly used to identify a risk category for a patient based on these tests.
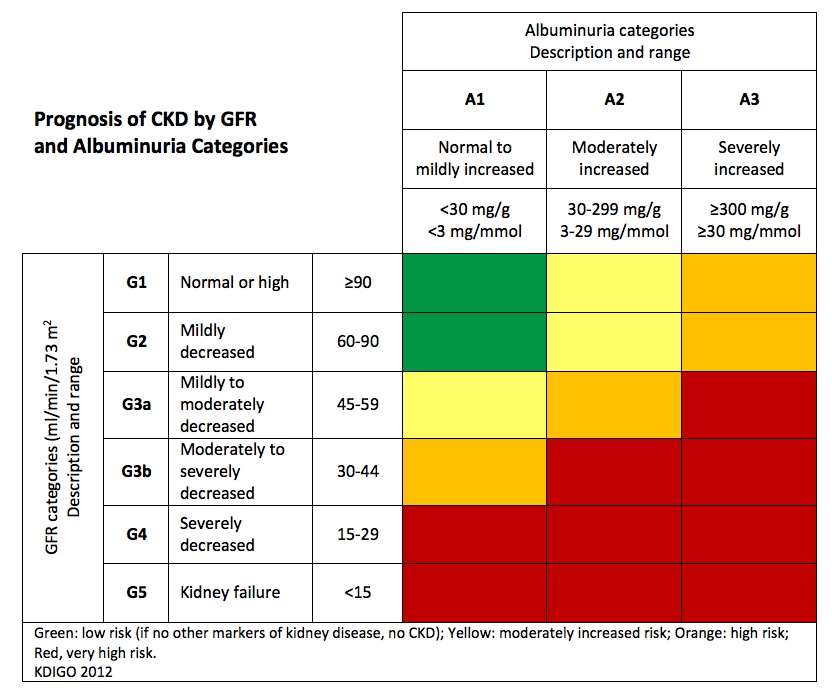
Source: National Kidney Foundation
To get this score, you must have a quantitative value for both blood and urine tests. The KED measure aligns with this guidance, and therefore a semi-quantitative test cannot be used to achieve this risk categorization. Because of that limitation, a semi-quantitative test alone is not considered best practice, and is not included in the quality measure.
Why did the 2023 update remove LOINC code 32294-1 from the KED measure specifications?
LOINC is a code system used to identify laboratory observations. When the KED measure was introduced, guidelines supported annual testing with both quantitative blood and quantitative urine tests for adults living with diabetes. Because of the quantitative test requirement, NCQA included in the measure specifications a list of LOINC test codes associated with quantitative urine tests.
NCQA discovered that a LOINC code (32294-1) may have been identified as a quantitative test when tests being coded to it were actually semi-quantitative. NCQA sought clarification from Regenstreif Institute staff and examined the use of the code, determining that it was not, in fact, being used to code a quantitative test. To ensure the integrity and consistency of the KED measure, in August 2023 NCQA updated MY 2024 specifications to remove that code.
Does the current measure prevent semi-quantitative testing?
No. The current measure does not prevent or discourage semi-quantitative testing. Patients or doctors may opt to use semi-quantitative tests for many reasons. However, if a semi-quantitative test is used and it is abnormal, guidelines recommend that the result be followed by a quantitative test processed by an accredited laboratory.
What is NCQA’s position on semi-quantitative tests?
NCQA supports early evidence-based detection and prevention of many conditions. The role of semi-quantitative testing in managing kidney disease is still being defined. Such testing may be important for screening, detection and prevention, but determining whether, and how, it should be used is a decision to be made by patients, ideally in consultation with physicians or other health professionals.
Does NCQA have any plans for semi-quantitative tests?
Cardiovascular-kidney-metabolic conditions are a priority area for NCQA to develop new types of measures and programs, including those aimed at early detection, risk reduction and prevention. NCQA welcomes inquiries from potential collaborating organizations, and from organizations that may be willing to fund development of new measures that cover all aspects of the quality of care, access, equity and experience across the patient journey.
In keeping with NCQA’s role as an independent organization that manages conflicts of interest, NCQA does not accept funding from organizations with commercial interests to develop quality measures. NCQA will also convene an independent expert panel to help us review updates to current evidence and guidelines, along with opportunities for new measures related to cardiovascular, kidney, and metabolic (CKM) syndrome.
What is NCQA, and what does it do?
NCQA is an independent, non-profit organization dedicated to improving the quality of health care in the United States. Patients are and always will be at the center of everything we do. NCQA was founded to improve quality and trust in health care and, for nearly 35 years, we have operated as a trusted resource for health plans, providers and patients to measure and improve the delivery of evidence-based care. As part of this work, NCQA develops and maintains HEDIS, a set of evidence-based quality measures that assess how well organizations are delivering and managing care.
HEDIS measures are trusted because NCQA uses a rigorous process involving careful review of evidence that supports the need for each measure; testing and evaluation of measure specifications; review and feedback from panels of nationally recognized experts; and voting by a multi-stakeholder panel of leaders representing a wide range of perspectives, including patients and caregivers, clinicians, insurers and government agencies.
NCQA panel members are vetted for conflicts of interest and industry influence to ensure that HEDIS measures reflect the best available science, align with public interests and take an unbiased view of measures’ importance, scientific soundness, feasibility and usability.
What NCQA does
- Helps identify care that represents evidence-based best practices (“high-quality care”) via experts, stakeholder engagement and public feedback, and careful consideration of research from existing bodies of evidence and clinical guidelines.
- Leverages our knowledge of high-quality care by creating standards and measures to help organizations improve their quality by evaluating how they perform over time and compared to other organizations.
- Uses those standards and measures in evaluation and benchmarking programs to help the public identify organizations that provide high-quality care and help build trust in health care.
What NCQA does not do
- NCQA is not a government agency and does not approve or deny tests for use.
- NCQA does not have regulatory oversight or decision-making authority over how care is covered/reimbursed.
- NCQA does not create clinical practice guidelines. Quality measures are tools for evaluation and are not designed to guide or replace professional judgement. Instead, NCQA identifies and specifies quality measures based on thorough research of existing guidelines and engagement with experts and stakeholder groups.
- NCQA does not rely on a single guideline or stakeholder to inform HEDIS measures. NCQA convenes broad sets of stakeholders, including patient advocacy organizations, to inform measures. NCQA considers a large body of scientific evidence and stakeholder feedback, including public comment.
Questions?
If you have any questions about the KED measure, or any other measure, submit them through My NCQA, and one of our subject matter experts will respond.








